The Epigenetic Aspect of Chronic Disease: Chemical Drivers, Transgenerational Persistence, and the Frontier of Epigenetic Editing
The landscape of public health in 2026 is defined by a fundamental re-evaluation of the etiology of chronic disease. For decades, the medical establishment operated under a paradigm that prioritized genetic determinism and individual lifestyle choices as the primary determinants of health. However, as the burden of chronic conditions—ranging from Type 2 diabetes and obesity to neurodegenerative disorders and infertility—reaches unprecedented levels, a new consensus has emerged. Current data indicates that synthetic chemical exposure is not merely an auxiliary risk factor but is increasingly recognized as a key driver, and in some specific contexts, the primary cause of the global chronic disease crisis.1 This transformation in understanding is rooted in the maturation of environmental epigenetics, a field that has demonstrated how chemical substances do not necessarily need to mutate the DNA sequence to cause lasting pathological changes. Instead, they trigger epigenetic modifications—chemical "tags" on the genome—that alter gene expression and, most alarmingly, can be transmitted across several generations.3
The 2026 Chemical Landscape: From Acute Toxicity to Epigenetic Drivers
The 2026 regulatory and scientific environment is grappling with the realization that the American population is undergoing a continuous, low-dose exposure to a cocktail of synthetic chemicals. The "Make America Healthy Again" (MAHA) Commission, established by Executive Order 14212 in mid-2025, has been instrumental in synthesizing this data for federal policy. Its inaugural and subsequent reports identify chemical exposure as one of the four main drivers of chronic disease in the United States, alongside poor nutrition, lack of physical activity, and chronic stress.2 The MAHA Strategy Report, presented in late 2025, specifically highlights the vulnerability of children, noting that over 60% of their calories come from highly processed foods, which serve as primary delivery vehicles for unhealthy additives and environmental contaminants.1
The scale of this exposure is quantified in the 2026 "Terrible Toxics" situation report from the Environmental Protection Network (EPN). This report identifies 12 high-risk pollutants where federal safeguards have been weakened or delayed, leading to widespread population exposure through food, water, and air.5 The data suggests a "multiplier effect" where families are not exposed to these pollutants in isolation but in combination, leading to synergistic health impacts that are often overlooked in single-chemical toxicity assessments.5
Population Exposure Scale to High-Risk Pollutants (2026)
Pollutant Category | Impacted Population / Scope | Primary Health Concerns and Links |
PFAS (Forever Chemicals) | 172 million people in drinking water | Hormone disruption, infertility, low birth weight, cancer 5 |
Lead (Water Systems) | 185 million people | Cognitive impairment, Parkinson’s disease, developmental delays 5 |
Lead Paint Hazards | 34.6 million homes | Childhood learning challenges, neurotoxicity 5 |
Ozone (Unhealthy Levels) | 125 million+ people | Respiratory distress, chronic asthma, immune dysfunction 5 |
Soot (PM 2.5 Pollution) | 85 million people | Cardiovascular disease, emergency room visits for respiratory failure 5 |
Mercury Contamination | All 50 States (Fish Advisories) | Neurodevelopmental issues, metabolic disruption 5 |
Microplastics | Near-universal (Consumer products) | Endocrine disruption, textile-based inhalation 2 |
The implications of this data extend beyond immediate toxicity. The shift in 2026 is toward understanding the "cumulative exposure" framework. Federal agencies, including the EPA, USDA, and NIH, are now mandated to develop research frameworks for cumulative exposure across chemical classes, prioritizing New Approach Methodologies (NAMs) such as organoids and computational simulations to bypass the limitations of traditional animal studies.1 This is a response to the realization that chemical residues pose environmental hazards that can manifest as chronic conditions decades after the initial exposure.7
Major Categories of Chemically-Induced Chronic Diseases
The link between chemical exposure and chronic disease is multifaceted, involving multiple organ systems and physiological pathways. In 2026, the focus has shifted toward categorizing these diseases based on their epigenetic and metabolic signatures.
Metabolic and Endocrine Disorders
Metabolic health is the most visible victim of widespread chemical exposure. The FDA's Human Foods Program (HFP) 2026 deliverables prioritize the reassessment of marketed food chemicals like phthalates, propylparaben, and synthetic antioxidants such as butylated hydroxyanisole (BHA) and butylated hydroxytoluene (BHT).6 These substances are recognized as "obesogens"—chemicals that disrupt the hormonal control of hunger and fat storage.
The role of epigenetics in these conditions is well-documented. For instance, DNA methylation patterns of the 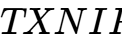 and
and 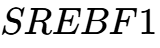 genes are now established biomarkers for Type 2 Diabetes (T2D) and Non-alcoholic fatty liver disease (NAFLD).3 In 2026, research presented at the Society of Toxicology (SOT) demonstrated that early-life exposure to toxicants like bisphenol A (BPA) and tributyltin (TBT) causes lasting epigenomic reprogramming that disrupts adult liver gene expression, effectively "pre-programming" the individual for metabolic failure.8 This is further complicated by the consumption of ultra-processed foods (UPFs), which the NIH and FDA are studying to understand how additives affect metabolic health and insulin sensitivity through epigenetic mechanisms.6
genes are now established biomarkers for Type 2 Diabetes (T2D) and Non-alcoholic fatty liver disease (NAFLD).3 In 2026, research presented at the Society of Toxicology (SOT) demonstrated that early-life exposure to toxicants like bisphenol A (BPA) and tributyltin (TBT) causes lasting epigenomic reprogramming that disrupts adult liver gene expression, effectively "pre-programming" the individual for metabolic failure.8 This is further complicated by the consumption of ultra-processed foods (UPFs), which the NIH and FDA are studying to understand how additives affect metabolic health and insulin sensitivity through epigenetic mechanisms.6
Neurodegenerative and Developmental Conditions
Chemical neurotoxicity is a primary driver of the rising rates of Parkinson’s disease, Alzheimer’s disease, and childhood learning challenges.5 The EPA and NIH are specifically investigating the impacts of air quality on children’s health, recognizing that soot and ozone exposure are linked to neuroinflammation.2
Epigenetic biomarkers for these conditions are becoming more precise. Aberrant DNA methylation and histone modifications are now used as early markers for Alzheimer’s and Parkinson’s, often detectable years before the onset of cognitive or motor decline.3 Research on the "Closer to Zero" initiative highlights that heavy metals like lead, arsenic, and cadmium in baby foods trigger sex- and tissue-specific alterations in gene regulation that disrupt placental function and early brain development.6
Reproductive Health and Infertility
Perhaps the most alarming trend in 2026 is the decline in reproductive health linked to chemical exposure. High-risk pollutants like PFAS and phthalates are directly connected to hormone disruption and infertility.5 In model organisms like C. elegans, exposure to common consumer product chemicals such as triclosan (TCS) and tetrabromobisphenol A (TBBPA) has been shown to alter the activities of histone methyltransferases and demethylases, leading to increased levels of repressive histone marks like H3K9me3 and H3K27me3.9 These changes are not just transient; they cause transgenerational developmental defects and reproductive toxicity that can persist long after the chemical is removed from the environment.9
Oncological Conditions
The field of cancer research in 2026 has increasingly moved toward "liquid biopsies" that detect cancer-specific DNA methylation patterns in blood or bodily fluids.3 Hypermethylation of tumor suppressor genes is now recognized as a primary hallmark of various cancers. For example, 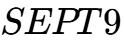 methylation is used for non-invasive colorectal cancer screening, while
methylation is used for non-invasive colorectal cancer screening, while 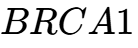 methylation serves as a prognostic marker for breast and ovarian cancer risk.3 The 2026 data emphasizes that environmental carcinogens like benzene, formaldehyde, and vinyl chloride drive these epigenetic alterations, which can then be inherited or acquired early in life, leading to a "multi-hit" model of oncogenesis where the first hit is often an environmental epigenetic mark.5
methylation serves as a prognostic marker for breast and ovarian cancer risk.3 The 2026 data emphasizes that environmental carcinogens like benzene, formaldehyde, and vinyl chloride drive these epigenetic alterations, which can then be inherited or acquired early in life, leading to a "multi-hit" model of oncogenesis where the first hit is often an environmental epigenetic mark.5
The 20-Generation Effect: Transgenerational Epigenetic Inheritance
One of the most profound discoveries emphasized in 2026 research is the "20-Generation Effect." This concept describes how certain epigenetic information can be transmitted between generations without any changes to the underlying DNA sequence, effectively creating a "cellular memory" of ancestral chemical exposure.4 While human longitudinal studies of this length are difficult, model organisms have provided a clear window into this phenomenon.
Mechanics of Transgenerational Inheritance
Research in Caenorhabditis elegans (nematodes) has identified several molecular mechanisms capable of transmitting this information. These include small non-coding RNA species (specifically 22G-RNAs), histone post-translational modifications, and the methylation of specific bases like 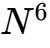 -adenine (6mA).4 Studies have shown that when these organisms are exposed to stressors or specific chemical agents, the resulting "epimutations" can propagate through a population in a manner reminiscent of DNA mutations.4
-adenine (6mA).4 Studies have shown that when these organisms are exposed to stressors or specific chemical agents, the resulting "epimutations" can propagate through a population in a manner reminiscent of DNA mutations.4
Data from 20-generation studies indicates that while many epimutations are short-lived (lasting 3-5 generations), a significant subset persists for the entire 20-generation duration.4 For instance, the loss of certain histone demethylases (like spr-5) leads to a transgenerational increase in 6mA levels that accumulates over 20 generations, resulting in progressively declining fertility and altered phenotypes in later generations.11
Longitudinal Impacts of Toxicants Across 20 Generations
Organism | Exposure / Agent | Duration | Observed Transgenerational Impact |
C. elegans | Heavy Metals (Arsenic) | 20 Generations | Foot and soft tissue damage patterns, gangrenous conditions 7 |
C. elegans | Nanoplastics (NH2-PS-NP) | Multiple Generations | Severe transgenerational neurotoxicity, locomotion defects 9 |
Bracon hebetor | Laboratory Stressors | 20 Generations | Significant shifts in biological efficacy and environmental safety markers 7 |
Medaka Fish | Bisphenol A (BPA) | Transgenerational | Transgenerational NAFLD and associated molecular changes 8 |
This 20-generation perspective suggests that the chronic disease crisis of 2026 may be, in part, the result of chemical exposures that occurred during the mid-20th century. The environmental stimulus—whether it was the introduction of synthetic pesticides or the widespread use of leaded gasoline—triggered epimutations that are only now reaching their full pathological potential in current generations. This "evolutionary drift" caused by epimutations can alter gene expression patterns across thousands of individuals without a single change in the genetic code.4
Breakthroughs in Epigenetics Research: The Path to Restoration
The realization that epigenetic changes are reversible is the cornerstone of 2026's most promising therapeutic breakthroughs. Unlike genetic mutations, which generally require permanent genome editing, epigenetic "tags" can be added, removed, or silenced using targeted molecular tools.
The Third Generation of CRISPR: Epigenetic Editing
In 2026, the biotech industry has moved beyond the first generation of CRISPR (which cut DNA) and the second generation (base editing) to the "Third Generation": Epigenetic Editing.13 This technology uses a nuclease-inactivated Cas protein (dCas) fused to epigenetic effector domains. Rather than changing the DNA sequence, these tools add or remove methyl groups or modify histones to turn genes on or off.13
A primary example is Scribe Therapeutics' STX-1150, which is projected to enter clinical trials in mid-2026. This therapy is designed to epigenetically silence the 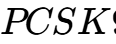 gene in the liver, providing a durable, long-term reduction in LDL cholesterol without permanently modifying the patient's DNA.14 Preclinical data in non-human primates showed a >50% LDL-C reduction that has been sustained for over 18 months from a single administration.14 This represents a shift from "pills for life" to "durable epigenetic interventions."
gene in the liver, providing a durable, long-term reduction in LDL cholesterol without permanently modifying the patient's DNA.14 Preclinical data in non-human primates showed a >50% LDL-C reduction that has been sustained for over 18 months from a single administration.14 This represents a shift from "pills for life" to "durable epigenetic interventions."
Epigenetic Reactivation and Disease Reversal
Another breakthrough involves switching on genes that have been silenced by disease or environmental toxins. Researchers have used epigenetic editing to remove methyl groups from the fetal globin gene, reactivating its expression to treat Sickle Cell disease.13 By lifting the "molecular brakes" on these silenced genes, doctors can provide a functional cure by restoring healthy blood cell production.13
In 2026, there is also emerging evidence that "epi-drugs"—small molecules that inhibit epigenetic "writers," "erasers," or "readers"—can reverse environmentally induced damage. For example, treatment with weak epigenetic modifiers has been shown to reverse BPA-induced transgenerational NAFLD in medaka fish, correcting the molecular changes that would have otherwise plagued future generations.8
2026 Epigenetic Therapeutic Pipeline
Therapy / Asset | Mechanism of Action | Target Condition | Stage (2026) |
STX-1150 (Scribe) | Epigenetic Silencing (ELXR) | Hypercholesterolemia | First-in-human (Mid-2026) 14 |
STX-1150 Prototype | dCasX-effector LNP | LDL-C Lowering | 18-month NHP durability 14 |
Pelabresib (CPI-0610) | BET Inhibitor | Myelofibrosis | Phase III trials 3 |
Ziftomenib (KO-539) | Menin-KMT2A Inhibitor | NPM1-mutant AML | FDA Breakthrough Therapy 3 |
Apabetalone (RVX-208) | Selective BET Inhibitor | End-stage Kidney Disease | Phase I/II 3 |
Larsucosterol (DUR-928) | Epigenetic Regulator | Alcoholic Hepatitis | Phase II 3 |
Modeyso (Dordaviprone) | H3K27M-mutant Targeting | Diffuse Midline Glioma | FDA Accelerated Approval 3 |
STAC-BBB Platform | Neurology Epigenetic Editing | Parkinson’s, ALS, Huntington’s | Early-stage partnerships 15 |
The focus in 2026 is on making these therapies more specific. While early epi-drugs often had genome-wide effects and potential toxicity, the new generation of epigenetic editors (like Scribe's ELXR technology) incorporates allosteric regulatory domains that reduce off-target effects and enhance on-target repression by four-fold.14 This high level of specificity is essential for treating chronic diseases that progress over decades.
Forecast and Probabilities: The 2036 Horizon
As the medical community looks toward the next ten years, the question is how many of these chemically-linked chronic conditions can be effectively cured or treated. The survey of 1,430 researchers working on rare and genetic diseases offers a benchmark: most expect genetic and epigenetic therapies to be the standard of care for many conditions before 2036.16
However, the "unconditional probability of premature death from NCDs" is only decreasing slowly on a global scale. At the current pace, it would take approximately 45 years to reach the Sustainable Development Goal of a one-third reduction.17 The 2026 data emphasizes that while the technology to cure these diseases is arriving, the exposure levels are still rising, creating a tug-of-war between biomedical progress and environmental degradation.5
10-Year Probability Matrix for Curability and Treatability (by 2036)
Disease Category | Probability of Major Treatment (Manageable) | Probability of "Cure" (Functional Resolution) | Key Enabling Breakthrough |
Hypercholesterolemia | 95% | 75% | Durable Epigenetic Silencing of |
Sickle Cell / Hemoglobinopathy | 90% | 80% | CRISPR Epigenetic Reactivation 13 |
Hematological Cancers (AML) | 85% | 45% | Targeted Menin-KMT2A Inhibitors 3 |
Type 2 Diabetes | 70% | 25% | Adipogenesis / |
Rare Neurogenetic Diseases | 65% | 30% | STAC-BBB Capsid Delivery Systems 15 |
Parkinson’s Disease | 55% | 15% | Alpha-synuclein Epigenetic Silencing 3 |
Chronic Kidney Disease | 60% | 20% | BET Bromodomain Inhibition 3 |
Chemical Infertility | 45% | 10% | Transgenerational Histone Resetting 8 |
These probabilities are influenced by the regulatory environment. The 2026 Forecast suggests that while litigation and "Loper Bright" upheaval may cause some delays, the move toward NAMs and away from animal testing will eventually streamline the path for these advanced therapies.2 Additionally, the use of AI in clinical trial data analysis and back-office operations is expected to accelerate the regulatory path to drug approvals throughout 2026 and beyond.21
Synthesis and Strategic Implications
The 2026 data presents a dual narrative of crisis and opportunity. The "Terrible Toxics" identified by the EPN and the chronic disease drivers highlighted by the MAHA Commission confirm that chemical exposure has fundamentally compromised the health of the American population.1 The discovery of the 20-generation effect underscores the staggering scale of this impact, suggesting that our current biological states are the products of an inherited toxicological legacy.4
However, the rapid transition of epigenetics from discovery to clinical application—a field now projected to be a $6.77 billion market by 2033—suggests a pathway to restoration.3 The emergence of "Operation Stork Speed" to protect infant nutrition and the FDA's "Closer to Zero" initiative for heavy metals represent the first steps in a grander strategy to decouple chemical exposure from chronic disease.6
For professional health stakeholders, the implication is a shift toward "restorative epigenetics." This involves not just the prescription of epi-drugs but the implementation of a comprehensive "Make Our Children Healthy Again" framework that integrates nutrition coaching, environmental safeguarding, and targeted epigenetic interventions.2 The goal is to move from managing the symptoms of chemically-induced disease to resetting the epigenetic clock of the population.
Nuanced Outlook on Restorative Medicine
The ability to treat or cure these conditions by 2036 will likely be determined by three factors: the precision of delivery systems (such as liver-targeted LNPs or BBB-crossing capsids), the regulatory authority to mandate the removal of toxic drivers, and the societal will to address the cumulative burden of pollutants. While the "20-generation" inheritance mechanism explains the persistence of chronic disease, the intrinsic reversibility of epigenetic marks provides the technical mechanism for its eradication. The next decade will determine whether medicine can scale these high-tech interventions fast enough to outpace the physiological decline caused by the "Terrible Toxics" of the modern era.5
By 2036, the integration of multi-omics data, AI-driven diagnostics, and 3rd-generation CRISPR tools will likely transform the most common chronic conditions from "unavoidable consequences of modern life" into "preventable and reversible epigenetic states".22 The path forward requires a unified approach that treats the environment and the epigenome as two sides of the same biological ledger.
Works cited
- The-MAHA-Strategy-WH.pdf - The White House, accessed March 22, 2026, https://www.whitehouse.gov/wp-content/uploads/2025/09/The-MAHA-Strategy-WH.pdf
- MAHA Commission Report Details Federal Response to Childhood ..., accessed March 22, 2026, https://www.hklaw.com/en/insights/publications/2025/09/maha-commission-report-details-federal-response
- The future of epigenetics: Emerging technologies and clinical ... - CAS, accessed March 22, 2026, https://www.cas.org/resources/cas-insights/epigenetics-emerging-technologies
- Fluctuations in chromatin state at regulatory loci occur spontaneously under relaxed selection and are associated with epigenetically inherited variation in C. elegans gene expression - Our journal portfolio - PLOS, accessed March 22, 2026, https://journals.plos.org/plosgenetics/article?id=10.1371/journal.pgen.1010647
- Health Experts Warn Americans Face Toxic Risks as EPA Abandons Health Protections, accessed March 22, 2026, https://www.environmentalprotectionnetwork.org/20260225_toxics-report-release/
- Human Foods Program 2026 Priority Deliverables | FDA, accessed March 22, 2026, https://www.fda.gov/about-fda/human-foods-program/human-foods-program-2026-priority-deliverables
- International Conference - ResearchGate, accessed March 22, 2026, https://www.researchgate.net/profile/Mujtaba_Momin2/publication/333079602_Adoption_of_Learner-centered_Approach_-_A_method_to_enhance_Learning_Effectiveness_for_Sustainability_Education/links/5cda7a3e92851c4eab9d8561/Adoption-of-Learner-centered-Approach-A-method-to-enhance-Learning-Effectiveness-for-Sustainability-Education.pdf
- SOT 65th Annual Meeting and ToxExpo Program, accessed March 22, 2026, https://www.toxicology.org/pubs/docs/Prog/2026Program.pdf
- A review of transgenerational and multigenerational toxicology in the in vivo model animal Caenorhabditis elegans - ResearchGate, accessed March 22, 2026, https://www.researchgate.net/publication/361563163_A_review_of_transgenerational_and_multigenerational_toxicology_in_the_in_vivo_model_animal_Caenorhabditis_elegans
- Fluctuations in chromatin state at regulatory loci occur ... - PMC - NIH, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC10013927/
- (PDF) DNA methylation on N-Adenine in C. elegans - ResearchGate, accessed March 22, 2026, https://www.researchgate.net/publication/275836112_DNA_methylation_on_N-Adenine_in_C_elegans
- 1 Fluctuations in chromatin state at regulatory loci occur spontaneously under relaxed 1 selection and are associated with epige - bioRxiv.org, accessed March 22, 2026, https://www.biorxiv.org/content/10.1101/2022.09.29.510125v1.full.pdf
- New CRISPR breakthrough could transform sickle cell treatment - Drug Target Review, accessed March 22, 2026, https://www.drugtargetreview.com/news/183354/new-crispr-breakthrough-could-transform-sickle-cell-treatment/
- Scribe → Scribe Therapeutics Projected to Enter the Clinic in Mid ..., accessed March 22, 2026, https://www.scribetx.com/news/scribe-therapeutics-projected-to-enter-the-clinic-in-mid-2026-with-stx-1150-a-pcsk9-targeting-crispr-epigenetic-silencing-therapy-for-durable-ldl-c-reduction
- sgmo-20231231 - SEC.gov, accessed March 22, 2026, https://www.sec.gov/Archives/edgar/data/1001233/000162828024010788/sgmo-20231231.htm
- Future of genetic therapies for rare genetic diseases: what to expect for the next 15 years? - PMC - NIH, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC10032453/
- Noncommunicable chronic diseases and health challenges in 2050 - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC12995342/
- 2026 Forecast for U.S. Federal and International Chemical Regulatory Policy - Bergeson & Campbell, P.C., accessed March 22, 2026, https://www.lawbc.com/wp-content/uploads/2026-Forecast.pdf
- CRISPR Therapeutics Highlights Strategic Priorities and Anticipated 2026 Milestones, accessed March 22, 2026, https://www.biospace.com/press-releases/crispr-therapeutics-highlights-strategic-priorities-and-anticipated-2026-milestones
- Pathways to chronic disease detection and prediction: Mapping the potential of machine learning to the pathophysiological processes while navigating ethical challenges - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC11880127/
- Industry Leaders Predict Life Science Trends for 2026 | The Scientist, accessed March 22, 2026, https://www.the-scientist.com/industry-leaders-predict-life-science-trends-for-2026-73980
- 2026 Life Sciences Forecast: AI, Next-Gen Cell & Gene Therapy, and Policy Disruption, accessed March 22, 2026, https://www.advi.com/insight/2026-life-sciences-forecast-ai-next-gen-cell-gene-therapy-and-policy-disruption/
- Epigenetic biomarkers for disease susceptibility and preventative medicine - PubMed, accessed March 22, 2026, https://pubmed.ncbi.nlm.nih.gov/38176413/
- CRISPR-Cas9 Gene Editing: Curing Genetic Diseases by Inherited Epigenetic Modifications, accessed March 22, 2026, https://pubmed.ncbi.nlm.nih.gov/38560484/
